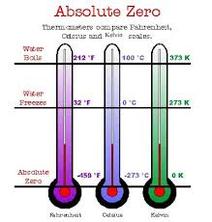
1) Absolute Zero: The temperature at which a substance has no kinetic energy per particle (thermal) to give up. This temperature corresponds to 0 K, or to -273c.
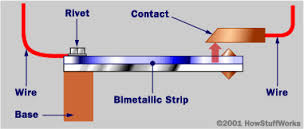
2) Bimetallic: It is a strip made of two different metals, one on each side. In a bimetal
strip, the two metals typically have different coefficients of expansion, so as
the temperature changes, the strip bends. Because of this, bimetal strips are
often used in devices such as thermostats.
strip, the two metals typically have different coefficients of expansion, so as
the temperature changes, the strip bends. Because of this, bimetal strips are
often used in devices such as thermostats.
3) calorie: Any of several approximately equal units of heat, each measured as the quantity
of heat required to raise the temperature of 1 gram of water by 1°C from a
standard initial temperature, especially from 3.98°C, 14.5°C, or 19.5°C, at 1
atmosphere pressure.
of heat required to raise the temperature of 1 gram of water by 1°C from a
standard initial temperature, especially from 3.98°C, 14.5°C, or 19.5°C, at 1
atmosphere pressure.
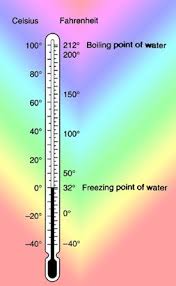
4) Celsius scale:a scale of temperature in which 0° represents the melting point of ice and 100°
represents the boiling point of water. See also centigrade Compare Fahrenheit scale
represents the boiling point of water. See also centigrade Compare Fahrenheit scale
5) Fahrenheit Scale: (Units) a scale of temperatures in which 32° represents
the melting point of ice and 212° represents the boiling point of pure water
under standard atmospheric pressure.
the melting point of ice and 212° represents the boiling point of pure water
under standard atmospheric pressure.
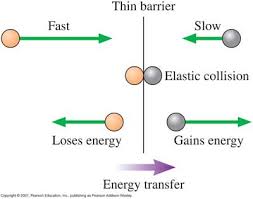
6) Heat: A form of energy associated with the motion of atoms or
molecules and capable of being transmitted through solid and fluid media by
conduction, through fluid media by convection, and through empty space by
radiation.
molecules and capable of being transmitted through solid and fluid media by
conduction, through fluid media by convection, and through empty space by
radiation.
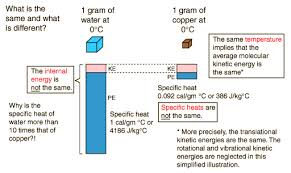
7) Internal Energy: The total kinetic and potential energy associated with the motions and relative
positions of the molecules of an object, excluding the kinetic or potential
energy of the object as a whole. An increase in internal energy results in a
rise in temperature or a change in phase.
positions of the molecules of an object, excluding the kinetic or potential
energy of the object as a whole. An increase in internal energy results in a
rise in temperature or a change in phase.
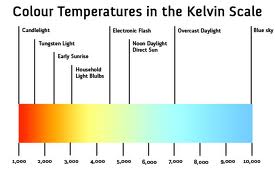
8) Kelvin Scale: A scale of temperature beginning at absolute zero (-273.15°C or -459.67°F). Each
degree, or kelvin, represents the same temperature increment as one degree on
the Celsius scale. On the Kelvin scale water freezes at 273.15 K and boils at
373.15 K.
degree, or kelvin, represents the same temperature increment as one degree on
the Celsius scale. On the Kelvin scale water freezes at 273.15 K and boils at
373.15 K.
9) Kilocalorie: A unit or heat. One kilocalorie equales 1000 calories, or the amount of heat required to raise the temperature of one kilogram of water by 1C.
10) Specific heat
Capacity: (General Physics) the heat required to raise unit mass of a substance
by unit
temperature interval under specified conditions, such as constant pressure:
usually measured in joules per kelvin per kilogram.
temperature interval under specified conditions, such as constant pressure:
usually measured in joules per kelvin per kilogram.
11)Temperature: A measure of the average kinetic energy of the particles in a sample of matter,
expressed in terms of units or degrees designated on a standard scale.
expressed in terms of units or degrees designated on a standard scale.
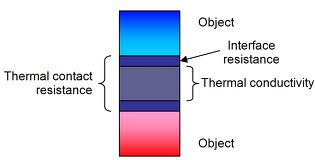
12)Thermal Contact: In heat transfer and thermodynamics, a thermodynamic system is said to be in thermal
contact with another system if it can exchange energy with it through the
process of heat. Perfect thermal isolation is an
idealization as real systems are always in thermal contact with their
environment to some extent.
contact with another system if it can exchange energy with it through the
process of heat. Perfect thermal isolation is an
idealization as real systems are always in thermal contact with their
environment to some extent.
13)Thermal Equilibrium: The state of two or more objects or substances in thermal contact when they have reached a common temperature
14)Thermotast: A type of valve or switch that responds to changes in temperature and its used to control the temperature of something.